Digestive Health Month: The changing landscape of digestive diagnostics
May is the World Digestive Health Month. It is a fitting time to take stock of how common gastrointestinal conditions truly are, how many go unrecognized, and how much the diagnostic landscape has changed in recent years.
Digestive diseases span a wide spectrum: bacterial infections that silently damage the stomach lining, autoimmune conditions that erode the gut’s ability to absorb nutrients, and chronic inflammatory diseases that can severely affect quality of life. Many of these conditions are often diagnosed late because symptoms may be absent, non-specific, or attributed to something else.
1. Helicobater pylori infection and atrophic gastritis
Helicobacter pylori is one of the most common chronic bacterial infections on earth. A 2023 meta-analysis found that global prevalence has declined from 58% in the 1980s to approximately 43% in recent years. Despite this downward trend, more than 40% of the world’s adult population is estimated to carry the bacterium. (1)
Most carriers have no symptoms. H. pylori quietly colonizes the stomach lining and, over years or decades, can drive a progression from chronic gastritis to atrophic gastritis (AG) and ultimately to gastric cancer. H. pylori has been classified as a Group I carcinogen.
Furthermore, a 2025 study based on 6.8 million Koreans shows that H. pylori infection is the dominant causal risk factor for gastric cancer, acting through intermediate stages including atrophic gastritis, intestinal metaplasia, and adenoma (2).
Atrophic gastritis (AG) is the gradual loss of the stomach’s specialized glandular cells and a precancerous condition for gastric adenocarcinoma, the most common type of gastric cancer. AG develops through two distinct pathways, each with its own clinical significance (H. pylori infection and autoimmune gastritis). Long-standing H. pylori infection is the most common cause of AG worldwide.
In autoimmune gastritis, the immune system directly targets the stomach’s parietal cells. This form of AG is not driven by infection but by autoimmune mechanisms. The resulting loss of parietal cells leads to reduced hydrochloric acid production (hypochlorhydria or achlorhydria) and impaired secretion of intrinsic factor, which is essential for vitamin B12 absorption.
Autoimmune gastritis is therefore a cause of pernicious anemia (B12 deficiency) but is also associated with malabsorption of iron, calcium, zinc, and magnesium.
Both H. pylori–associated atrophic gastritis and autoimmune atrophic gastritis are associated with an increased risk of gastric adenocarcinoma. The GastroPanel® quick test NT test is intended to support the identification of atrophic changes in the gastric corpus based on its biomarker profile. Reduced pepsinogen I levels together with a decreased PGI/PGII ratio are indicative of corpus atrophy, irrespective of the underlying etiology. The result of the H. pylori biomarker may assist in differentiating between H. pylori–associated and autoimmune pathways.
Read more: Is there a link between atrophic gastritis and osteoporosis?
How is H. pylori infection diagnosed?
First-line diagnosis
Non-invasive diagnostic methods such as blood-based panels can provide valuable information and help determine whether further investigation is necessary. This is where GastroPanel® quick test NT plays a central role.
GastroPanel® quick test NT is a blood-based panel that measures four biomarkers from a single sample: pepsinogen I, pepsinogen II, gastrin-17, and H. pylori antibodies. Together, they detect not only H. pylori infection, but also whether chronic infection has progressed to atrophic gastritis.
It enables first line diagnosis of dyspeptic patients and screening of both symptomatic and asymptomatic patients, to help select patients for further investigation.
Read more: Chile adopts GastroPanel® technology in national cancer prevention strategy
Diagnosis during gastroscopy
Rapid confirmation of H. pylori status is valuable when a patient undergoes gastroscopy. With BIOHIT H. pylori UFT300 quick test, a biopsy can be tested immediately to diagnose H. pylori infection. The rapid urease test delivers a result from a biopsy specimen in as little as 5 minutes, enabling diagnosis and reporting in a single appointment.
Triage tool for prioritization
Gastroscopy remains the gold standard for directly visualizing and biopsying the gastric mucosa. However, it is invasive and resource-intensive. Furthermore, many healthcare systems are burdened by long waiting times for endoscopies, even though many referred patients have no organic disease.
GastroPanel® quick test NT enables a non-invasive approach first: patients with normal biomarker profiles can be managed without endoscopy, while those with markers suggesting atrophy, H. pylori infection, or acid secretion abnormalities can be prioritized for gastroscopy. This channels resources toward patients who are most likely to benefit from an endoscopy.
A prospective UK validation study of 268 dyspepsia patients confirmed that GastroPanel® can reliably distinguish patients who can be safely managed without gastroscopy from those with moderate-to-severe corpus atrophy in need of further investigation (3).
Read more: How does GastroPanel® perform as a triage system for gastric cancer risk assessment?
2. Inflammatory bowel disease (IBD)
Chronic abdominal pain, bloating, diarrhea, and fatigue are among the most common reasons for gastroenterology referrals. However, these are common symptoms characteristic of several conditions with different causes and trajectories including irritable bowel syndrome (IBS), Crohn’s disease and ulcerative colitis (Inflammatory bowel disease, IBD).
Misclassifying IBD as IBS delays treatment and allows disease progression. Conversely, subjecting IBS patients to invasive investigations wastes resources and causes unnecessary burden on the patient. Reliable biomarkers that distinguish organic inflammation from functional symptoms are therefore essential.
Using diagnostics to differentiate IBD from IBS
Fecal calprotectin is a protein released by neutrophils during intestinal inflammation. Its concentration in stool correlates closely with the degree of mucosal inflammation. Therefore, fecal calprotectin is a well-validated tool for differentiating IBD from IBS.
BIOHIT Calprotectin is a quantitative test that reliably differentiates between IBD and IBS from a stool sample. Beyond initial diagnosis, BIOHIT Calprotectin is also valuable for monitoring disease activity in known IBD patients.
3. Celiac disease
Celiac disease is an autoimmune condition in which gluten ingestion triggers immune-mediated damage to the small intestinal mucosa in genetically predisposed individuals.
The majority of celiac disease cases go undetected as 83–95% of celiac patients in developed countries remain undiagnosed, and the figure is likely higher in developing countries. (4)
While classic symptoms such as diarrhea and weight loss are commonly associated with the condition, many patients may be asymptomatic or exhibit non-classical signs including fatigue, iron-deficiency anemia, and osteoporosis, which can lead to delayed or missed diagnoses.
Celiac disease diagnostics
Accurate diagnosis of celiac disease is essential, and tools like the BIOHIT Celiac quick test make this process much easier. This rapid test provides reliable results in just a few minutes, helping healthcare providers quickly identify celiac disease and allowing patients to receive appropriate care without unnecessary delays.
The BIOHIT Celiac quick test is a rapid immunochromatographic test that detects antibodies against tissue transglutaminase (anti-tTG; IgA, IgG, IgM) from a whole blood, plasma or serum sample – including fingertip whole blood samples – with results in ten minutes. It is designed for use in primary care settings, outpatient dietetic clinics, and pharmacies.
Read more: BIOHIT Celiac quick test
4. Colorectal cancer
Colorectal cancer (CRC) is one of the most common cancers worldwide: it is the third most commonly diagnosed cancer globally and one of the leading causes of cancer mortality (5).
Early detection of colorectal cancer (CRC) significantly improves survival rates, as patients diagnosed before symptoms appear have a much better prognosis. Fecal immunochemical testing (FIT) has been shown to effectively lower CRC mortality by enabling timely identification and intervention.
BIOHIT ColonView® quick test (FIT) is a lateral flow FIT test designed for the detection of fecal occult blood. The test is based on highly sensitive immunochemical detection of hemoglobin and hemoglobin/haptoglobin complex. It is used to screen for lower gastrointestinal (GI) pathologies, such as colorectal cancers and large adenomas that bleed.
Read more: BIOHIT ColonView® quick test (FIT)
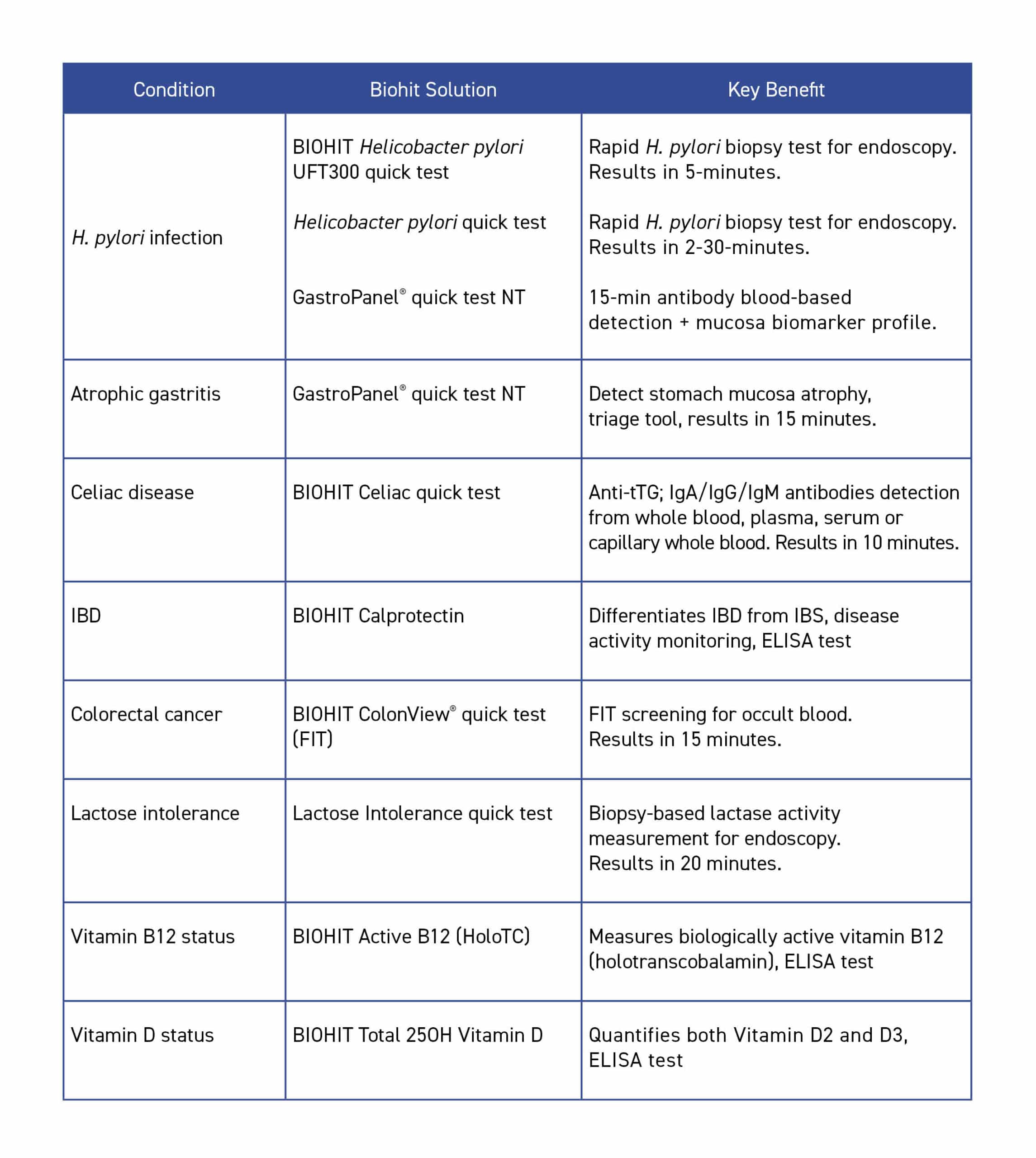
BIOHIT’s diagnostic toolkit for digestive health
Many of the most consequential gastrointestinal conditions, including H. pylori infection, atrophic gastritis, celiac disease, and early-stage colorectal cancer, are frequently present long before symptoms prompt investigation. Fortunately, the diagnostic tools to find them earlier are becoming faster, less invasive, and more accessible.
Biohit’s portfolio addresses several areas of diagnosing digestive diseases. The table below provides an overview of Biohit’s diagnostic solutions and their clinical applications.
References
- Li, et al. (2023). Global prevalence of Helicobacter pylori infection between 1980 and 2022: a systematic review and meta-analysis. The Lancet Gastroenterology & Hepatology, 8(6), 553–564. https://doi.org/10.1016/S2468-1253(23)00070-5
- Oh, et al. (2025). Quantifying the effects of the Correa pathway from Helicobacter pylori infection to gastric cancer: causal inference found in 6.8 million Koreans. BMC Cancer, 26(1), 174. https://doi.org/10.1186/s12885-025-15507-9
- Papadia, et al. (2025). Can GastroPanel be used as a triage tool to select patients with advanced atrophic gastritis for gastroscopy? A prospective clinical validation study. BMJ Open Gastroenterology, 12(1), e001559. https://doi.org/10.1136/bmjgast-2024-001559
- Singh, et al. (2018). Global Prevalence of Celiac Disease: Systematic Review and Meta-analysis. Clinical Gastroenterology and Hepatology, 16(6), 823–836. https://doi.org/10.1016/j.cgh.2017.06.037
- Cancer Today. https://gco.iarc.fr
