Colorectal Cancer Awareness Month: Why early detection of CRC matters
March marks Colorectal Cancer Awareness Month and gives much deserved attention to one of the most common cancers worldwide, accounting for nearly a million deaths every year. Colorectal cancer (CRC) is the third most commonly diagnosed cancer globally (10.2% of new cases) and one of the leading causes of cancer mortality (9.3% of cancer deaths) (1).
“Colorectal cancer (CRC) is the third most commonly diagnosed cancer globally (10,2 % of new cases) and one of the leading causes of cancer mortality (9,3% of cancer deaths).”
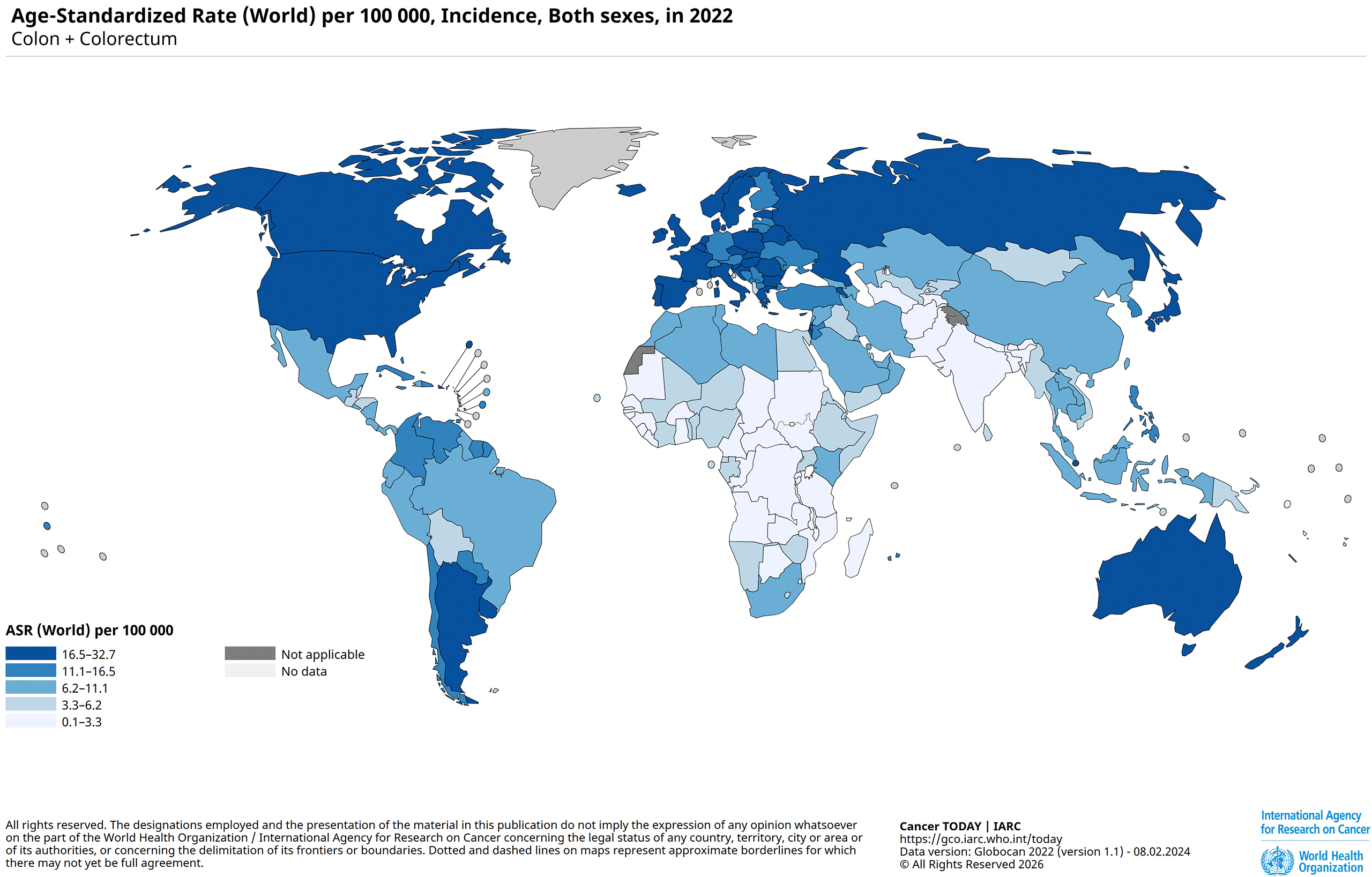
2022 figures from the International Agency for Research on Cancer (IARC) (2), a specialized cancer agency under the World Health Organization, show that each year there are about 1.9 million new cases of colorectal cancer and 900,000 deaths due to CRC worldwide.
Global burden: incidence and mortality vary by region
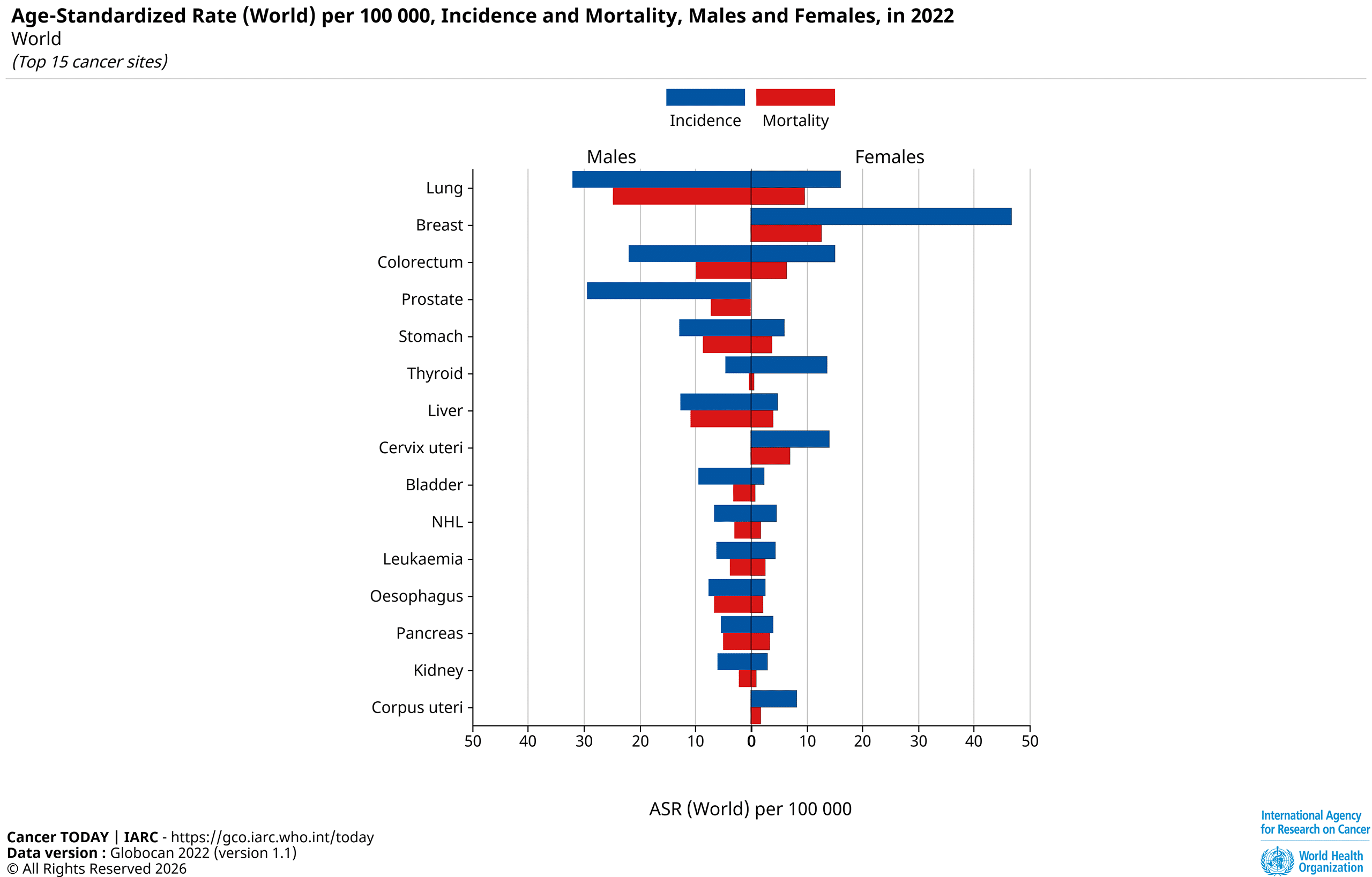
Globally, colorectal cancer is the third most common cancer for both sexes, with 1,07 million new cases in 2022 for men and 857,000 for women. For men, this accounts for 10.4% of new cancer cases and 8.9% for women. (1)
Incidence and mortality of colorectal cancer around the world, based on 2022 data (1):
Europe: 12.0% of all cancer cases and 12.5% cancer deaths.
- North America: 6.9% of all cancer cases and 9.4% cancer deaths.
- Latin America and the Caribbean: 9.4% of all cancer cases and 9.8% cancer deaths.
- Asia: 9.8% of all cancer cases and 8.5% cancer deaths.
These regional differences reflect variations in population age structure, lifestyle-related risk factors, screening coverage, and access to treatment.
Early detection is critical for CRC outcomes
Colorectal cancer can take over 10 years to develop from pre-malignant adenomas. As a result, the window for early cancer detection is long. Screening for CRC increases the rate of early-stage colorectal cancer detection and improves the survival rate.
A study examining colon and rectal cancer survival in seven high-income countries (Australia, Canada, Denmark, Ireland, New Zealand, Norway and the UK) found that more than 1 in 3 colon cancers are diagnosed at an early, localized stage (except in New Zealand). Meanwhile, the proportion of colon cancer cases presenting with distant disease ranges from 21.5% to 28.7% in different countries. (3)
“Although CRC is responsible for over 9 % of all cancer deaths, the survival rate of the disease has improved over the past 20 years (3).”
However, the number of deaths attributed to colorectal cancer is projected to increase in the coming years, even though the mortality rate is on a downward trend. The growth in death comes mainly as a result of growing and ageing populations. (3)
Data: Global Cancer Observatory (IARC), GLOBOCAN 2022. https://gco.iarc.who.int/today, accessed 6th March 2026.
Colorectal cancer is increasingly common among younger adults
IARC has stated that colorectal cancer incidence has been on the rise in people under 50 for at least 20 years. Yet the underlying causes behind this concerning figure are still unknown. (2)
The risk factors for colorectal cancer include:
- Old age
- Family history
- Male sex
- Familial adenomatous polyposis
- Lynch Syndrome
- Colorectal polyps
- Ulcerative colitis and Crohn’s disease
Common colorectal cancer symptoms include bloody stool, change in bowel habits, rectal or abdominal mass, sensation of need to defecate, unintentional weight loss, anemia and rectal or abdominal pain. These signs may be a cause for testing.
While symptomatic individuals are often investigated diagnostically, national screening programmes may test asymptomatic individuals, particularly those at elevated risk due to family history of CRC.
Evidence supports FIT-based screening of CRC
Studies support the use of fecal occult blood (FOB) testing as a first-line approach in colorectal cancer (CRC) screening. Immunochemical FOB tests (FIT/iFOB) have consistently outperformed guaiac-based FOB tests (gFOB) for colorectal cancer detection. (4)
In a systematic review and meta-analysis including 31 studies, FIT demonstrated a higher overall diagnostic performance for invasive colorectal cancer than gFOB. This supports the use of FIT as the preferred option when implementing new screening programmes. (4)
“For the combined endpoint of adenoma + carcinoma, BIOHIT ColonView® quick test achieved 97.2% sensitivity and 85.8%…”
In a colonoscopy-referral cohort (n=300) with biopsy-confirmed endpoints, Vasilyev et al. (5) compared the BIOHIT ColonView® quick test FIT test with gFOB. For the combined endpoint of adenoma + carcinoma, BIOHIT ColonView® quick test achieved 97.2% sensitivity and 85.8% specificity, which was substantially higher than the AUC of gFOBT (0.916 vs 0.774). (5)
The BIOHIT ColonView® quick test is based on dual-marker detection of fecal haemoglobin (Hb) and the hemoglobin/haptoglobin (Hb/Hp) complex. Because free hemoglobin can degrade during passage through the colon, the more stable Hb/Hp complex may improve detection of bleeding originating from the proximal colon.
Further analysis by Syrjänen et al. (6) of the Finnish CRC screening programme highlighted how screening design affects adenoma detection. Their study estimated performance under different strategies. Sensitivity for the detection of Adenoma was estimated to be very low (approximately 12.5%, AUC 0.560) under a one-sample hemoglobin-only approach, despite high CRC detection (CRC AUC 0.950). By contrast, adding Hb/Hp and using sequential sampling increased the sensitivity for adenoma detection to 47.5% (AUC 0.730). (6)
These results suggest that this new modality for testing supports stronger adenoma detection and the primary prevention aims of screening (detecting and removing precancerous lesions), in addition to secondary prevention (early cancer detection).
Read more about the BIOHIT ColonView® quick test (FIT) or download the brochure. ->
Biohit’s role in colorectal cancer diagnostics
Although CRC remains one of the leading causes of cancer mortality globally, the long development pathway from adenoma to carcinoma provides an opportunity for prevention.
Biohit develops diagnostic tools for gastrointestinal health, including the ColonView® FIT quick test, which is designed to support early detection of colorectal bleeding associated with CRC and large adenomas.
BIOHIT ColonView® quick test uses a dual-marker approach (Hb and Hb/Hp complex). This combination of two biomarkers makes ColonView® superior in detecting bleeding from the entire colorectal tract, including the proximal colon.
Benefits of ColonView®’s dual-marker approach include:
- Accurate results in approximately 15 minutes, making ColonView® suitable for both screening and diagnostic workflows in clinical settings.
- Improved detection of bleeding and accuracy for colorectal cancer screening.
- Resistance to acid and proteolytic degradation
- Stability over the entire course of the large bowel.
- Detection of both proximal and distal bleeds, minimizing the risk of missing lesions from the proximal colon.
- Improved performance compared to Hb alone
Want to learn more about ColonView® and our solutions?
Contact us and one of our experts will get back to you.
References:
- Cancer Today. https://gco.iarc.fr
- IARC marks Colorectal Cancer Awareness Month 2025. https://www.iarc.who.int/news-events/iarc-marks-colorectal-cancer-awareness-month-2025/
- Araghi, et. al. (2021). Colon and rectal cancer survival in seven high-income countries 2010-2014: variation by age and stage at diagnosis (the ICBP SURVMARK-2 project). Gut, 70(1), 114–126. https://doi.org/10.1136/gutjnl-2020-320625
- Meklin, et. al. (2020). Fecal Occult Blood Tests in Colorectal Cancer Screening: Systematic Review and Meta-analysis of Traditional and New-generation Fecal Immunochemical Tests. Anticancer research, 40(7), 3591–3604. https://doi.org/10.21873/anticanres.14349
- Vasilyev, et.al. (2015). A New-Generation Fecal Immunochemical Test (FIT) Is Superior to Quaiac-based Test in Detecting Colorectal Neoplasia Among Colonoscopy Referral Patients. Anticancer research, 35(5), 2873–2880.
- Vasilyev, et. al. (2015). A New-Generation Fecal Immunochemical Test (FIT) Is Superior to Quaiac-based Test in Detecting Colorectal Neoplasia Among Colonoscopy Referral Patients. Anticancer research, 35(5), 2873–2880
