GastroPanel® Unified ELISA
Laboratory-based stomach test
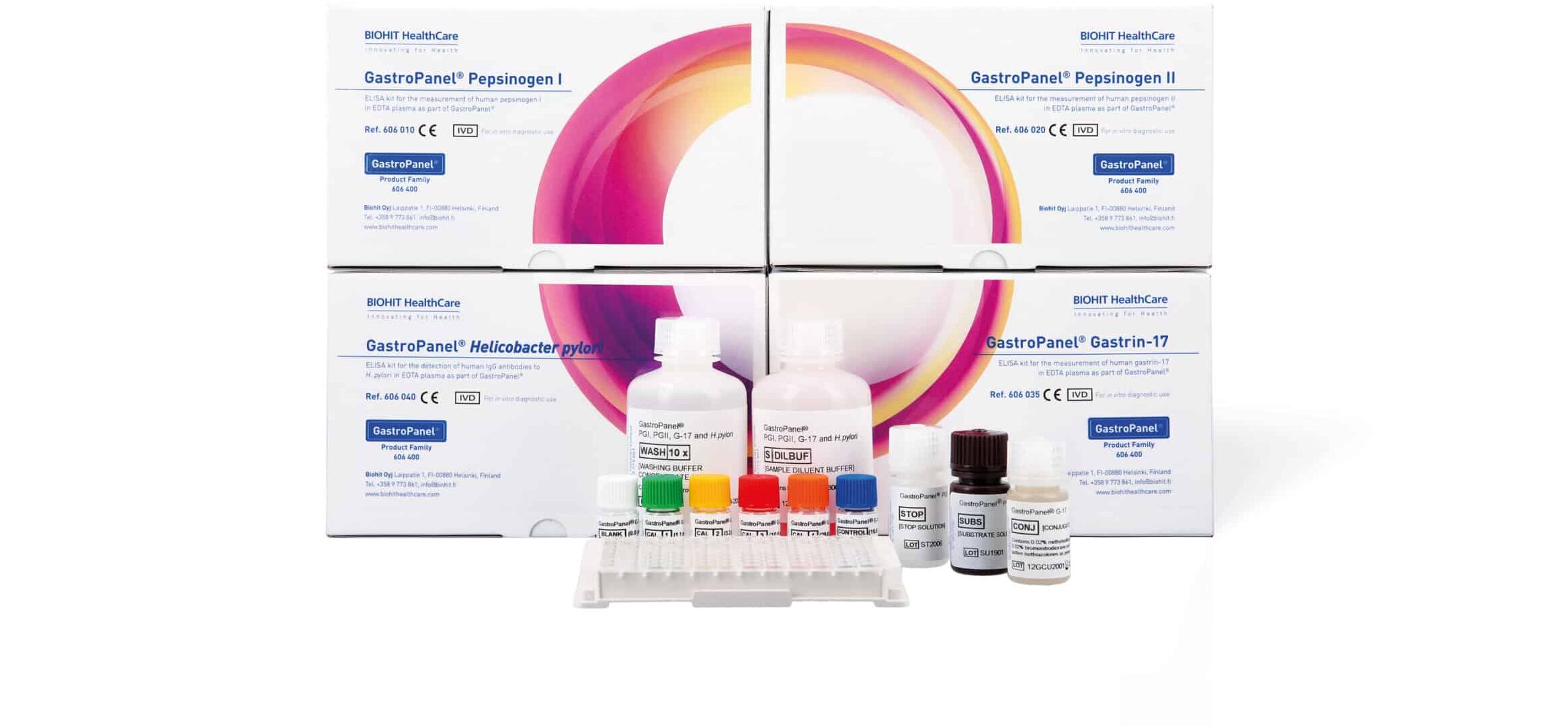
GastroPanel® Unified is a quantitative, non-invasive laboratory diagnostic test panel designed for the assessment of gastric mucosal structure and function. The test enables identification of Helicobacter pylori infection, atrophic gastritis, and abnormal gastric acid output, supporting efficient clinical decision-making and patient triage.
GastroPanel® Unified consist of four ELISA test kits to be used in combination. Each kit measures a clinically relevant gastric biomarker from a blood sample. It is intended for use as a first-line diagnostic and screening tool by healthcare professionals in laboratory.
A dedicated software GastroSoftTM can be used to interpret and report the combination of all four test results of GastroPanel Unified. GastroSoft is designed to assist clinician/general practitioner in interpreting GastroPanel Unified test results along side anamnestic information. The final responsibility of the diagnosis and treatment always rests with the patient’s doctor.
→ Talk to our experts about your needs
GastroPanel® Unified quantitatively measures:
Pepsinogen I (PGI): Marker of corpus mucosal integrity
Pepsinogen II (PGII): Inflammation marker of the entire gastric mucosa
Gastrin-17 (G-17): Marker of antral mucosal integrity, and acid secretion capacity
Antibodies against Helicobacter pylori: Marker to assess exposure to and infection with H. pylori.
Sample types
Venous whole blood
EDTA plasma
Key benefits
• Non-invasive and patient-friendly
• Quantitative results with interpretation report (GastroSoft® software)
• Supports efficient triage and referral
• Backed by extensive scientific and clinical research
Brochures and instructions for use
GastroPanel® Concept
GastroPanel® IFU
The GastroPanel® test panel consists of four assays (REF)
| REF | Product | Qty |
|---|---|---|
| 601300 | GastroPanel® | 1 package |
| 601010.01 | Pepsinogen I | 96 wells |
| 601020.02 | Pepsinogen II | 96 wells |
| 601035 | Gastrin-17 Advanced | 96 wells |
| 601040.02 | Helicobacter pylori IgG | 96 wells |
| REF | Product | Qty |
|---|---|---|
| 606400 | GastroPanel® Unified | 1 package |
| 606010 | GastroPanel® Pepsinogen I | 96 wells |
| 606020 | GastroPanel® Pepsinogen II | 96 wells |
| 606035 | GastroPanel® Gastrin-17 | 96 wells |
| 606040 | GastroPanel® Helicobacter pylori IgG | 96 wells |
Literature
Meta-analyses:
- Syrjänen 2022 | 49 studies, 22,597 patients: Accuracy of Serum Biomarker Panel (GastroPanel®) in the Diagnosis of Atrophic Gastritis of the Corpus. Systematic Review and Meta-analysis
- Zagari et al 2017 | 20 studies, 4241 patients: Systematic review with meta‐analysis: diagnostic performance of the combination of pepsinogen, gastrin‐17 and anti‐Helicobacter pylori antibodies serum assays for the diagnosis of atrophic gastritis
- Syrjänen 2016 | 27 studies, 8654 patients: A Panel of Serum Biomarkers (GastroPanel®) in Non-invasive Diagnosis of Atrophic Gastritis. Systematic Review and Meta-analysis
Some of the key articles:
- Koivurova et al 2020: Screening of the patients with autoimmune thyroid disease (AITD) and type 1 diabetes mellitus (DM1) for atrophic gastritis AG) by serological biomarker testing (GastroPanel®). EC Gastroenterol Digest Syst.
- Chapelle et al 2020: A panel of stomach-specific biomarkers (GastroPanel®) for the diagnosis of atrophic gastritis: A prospective, multicenter study in a low gastric cancer incidence area.
- Cai et al 2019: Development and validation of a prediction rule for estimating gastric cancer risk in the Chinese high-risk population: a nationwide multicentre study
- Syrjänen et al 2019: GastroPanel® Biomarker Assay: The Most Comprehensive Test for Helicobacter pylori Infection and Its Clinical Sequelae. A Critical Review
- Den Hollander et al 2019: Surveillance of premalignant gastric lesions: a multicentre prospective cohort study from low incidence regions
- Tepes et al 2018: Premalignant Gastric Lesions in Patients Included in National Colorectal Cancer Screening
- Tue et al 2017: A Serological Biopsy Using Five Stomach-Specific Circulating Biomarkers for Gastric Cancer Risk Assessment: A Multi-Phase Study
- Benberin et al 2013: Prevalence of H. pylori Infection and Atrophic Gastritis Among Symptomatic and Dyspeptic Adults in Kazakhstan. A Hospital-based Screening Study Using a Panel of Serum Biomarkers
- Agreus et al 2012: Rationale in diagnosis and screening of atrophic gastritis with stomach-specific plasma biomarkers
- Storskrubb et al 2008: Serum biomarkers provide an accurate method for diagnosis of atrophic gastritis in a general population: The Kalixanda study
Supported by the international consensus reports:
- Malfertheiner et al 2017: Management of Helicobacter pylori infection—the Maastricht V/Florence Consensus Report (see page 11)
- Kyoto 2015: Kyoto global consensus report on Helicobacter pylori gastritis (see page 9)
You can find more studies and abstracts from Literature search